5. PICO Questions, Evidence Review, and New Recommendations
PICO 1: Do pregnancy-related conditions (hypertensive disorders of pregnancy and other related complications) identify women at increased risk of premature CVD warranting lipid screening?
Pregnancy complications such as preeclampsia and related hypertensive disorders of pregnancy, gestational diabetes, placental abruption, preterm delivery, stillbirth, and delivery of a low birth weight infant are associated with a higher lifetime risk of developing CV risk factors (hypertension; type 2 diabetes mellitus; dyslipidemia, especially hypertriglyceridemia and low HDL-C; metabolic syndrome; and subclinical atherosclerosis) and overt ASCVD.[20],[21] The strongest and most abundant evidence linking pregnancy events and ASCVD is for preeclampsia, in which there is a twofold relative risk of developing premenopausal ASCVD, with onset at 10-15 years after delivery[20] compared with women who had uncomplicated pregnancies. This risk is highest if preeclampsia is recurrent (ie, 28% lifetime risk of ASCVD, or within 25 years after delivery[22]), or if associated with preterm delivery (before 37 weeks’ gestation) and other adverse conditions (chest pain, dyspnea, low platelet count, elevated liver enzymes, intrauterine growth restriction) or severe complications (eclampsia, stroke, myocardial ischemia, hepatic rupture, acute kidney injury with need for hemodialysis).[23] ASCVD risk is partly mediated by the development of chronic hypertension and metabolic syndrome.[24] There is often silent and subclinical endothelial dysfunction after hypertensive disorders of pregnancy suggesting accelerated vascular aging.[25],[26] National CV societies,[1],[27] including the CCS,[1] have recommended performing lipid and metabolic screening in postpartum women who have had these complications, although whether specific thresholds warranting pharmacotherapy differ from those typically used in the general population is not known. Although it is true that these women have a low absolute risk of ASCVD over the short term, the postpartum period might represent “a teachable moment” to engage young women in CV prevention and might result in long-term benefits through health behaviour interventions with or without pharmacological intervention. Treatment decisions should be guided on the basis of lifetime risk in conjunction with patient values and preferences.[28]
Recommendation
- Among women who have had a pregnancy complication such as hypertensive disorders of pregnancy, gestational diabetes, preterm birth, stillbirth, low birth weight infant, or placental abruption, we recommend screening with a complete lipid panel in the late postpartum period, because these women have a higher risk of premature CVD and stroke with onset 10-15 years after index delivery (Strong Recommendation; Moderate-Quality Evidence).
- We recommend counselling women who have any of these pregnancy-related complications of the increased lifetime risk of ASCVD, and reinforcing the importance of healthy behaviours (ie, maintaining a healthy body weight, 150 weekly minutes of moderate intensity aerobic physical activity, avoiding tobacco consumption, no more than moderate alcohol consumption, stress management, and adopting a healthy dietary pattern, such as the Mediterranean diet (Strong Recommendation; LowQuality Evidence).
- To assist with decisions about lipid-lowering pharmacotherapy in this patient population, we recommend favouring CV age, over 10-year risk calculators (Strong Recommendation; Low-Quality Evidence).
Values and Preferences
Although much of this observed risk among women who have had a pregnancy-related complication might be due to conventional ASCVD risk factors, complications such as preeclampsia might lead to ASCVD through accelerated vascular aging or other pathways warranting additional future research. There is insufficient evidence to guide decisions about use of lipid-lowering therapy in women on the basis of pregnancy factors alone. The American Heart Association 2019 CV prevention guidelines[27] consider preeclampsia a risk enhancer warranting early screening, healthy behaviour interventions, and possibly shifting of risk category from borderline to intermediate risk (ie, eligible for statin or other lipid-lowering therapy). We suggest individual discussions about statin or other lipid-lowering pharmacotherapy, considering each patient’s lifetime risk/individual risk factors along with severity and recurrence of pregnancy complications (in particular preterm preeclampsia with adverse conditions), balanced against the potential side effects and harms of long-term therapy. Although statins were previously considered teratogenic on the basis of earlier animal studies, this has not been consistently shown in recent human studies.[29],[30] A part of the observed increase in risk of congenital malformations might be due to underlying medical conditions rather than treatment with statin therapy itself.[29] Furthermore, there appears to be a differential effect on the basis of the type of compound, with most cases of congenital malformations being seen among infants whose mothers took lipophilic compounds (eg, atorvastatin, lovastatin, simvastatin) as opposed to hydrophilic compounds (eg, pravastatin, rosuvastatin).[31],[32] Therefore, in women who are reproductive age and who are eligible and considering statin therapy for ASCVD risk reduction on the basis of CV age or lifetime risk of ASCVD, we suggest the use of hydrophilic compounds over lipophilic compounds because of easier passage through the placenta with the latter molecules. It should be noted that for most reproductive women who take statin therapy for primary prevention of ASCVD, an effective birth control method is recommended with interruption of therapy before a planned pregnancy or at the time of an unplanned positive pregnancy test. These treatments can be resumed after delivery, when breastfeeding is completed. Referral to a specialist in obstetrical medicine or in fetal-maternal medicine should also be considered in the management of statin and nonstatin therapies in pregnant women or in women planning pregnancy.
PICO 2a: Is there evidence to promote non-HDL-C over ApoB or ApoB over non-HDL-C for screening and treatment purposes?
Previous versions of these guidelines have used LDL-C as the primary laboratory measurement for considering initiation of statin treatment and as a treatment target in low-, intermediate-, and high-risk individuals. Beginning with the 2012 guidelines, it has been recommended that non-HDL-C and ApoB could be used as alternate targets to LDL-C in any individual with triglyceride level > 1.5 mmol/L.[1],[33] The rationale for this is that above this level of triglyceride, some cholesterol in LDL particles is replaced by triglyceride, which promotes production of more atherogenic small dense LDL particles,[34] and makes the amount of cholesterol in LDL-C an unreliable reflection of LDL particle number.[35] In addition, other particles, such as remnants of chylomicrons and very LDL-C, as well as Lp(a), all accumulate in the artery wall and contribute to atherogenesis, whereas HDL-C does not. Therefore, estimation of the concentration of all atherogenic particles requires a broader focus than a measurement of LDL-C. Non-HDL-C (indirectly) and ApoB (directly) provide a more accurate assessment of the total concentration of atherogenic particles than LDL-C. Non-HDL-C and ApoB are, for this reason, both better predictors of CV event risk and benefit of lipid-lowering therapy compared with LDL-C.[36],[37] On the basis of these previous recommendations, non-HDL-C is now routinely reported across Canada at no additional cost, on the basis of the simple calculation of total cholesterol minus HDL-C. ApoB is also available as an insured laboratory test in all provinces except Ontario. Levels of non-HDL-C and ApoB are not significantly changed in the postprandial state in individuals with triglycerides < 4.5 mmol/L, whereas LDLC can be lowered by up to 10% because of triglyceride enrichment of LDL-C.[38],[39] After the guideline recommendation that was introduced in 2016 allowing for nonfasting collections for screening and follow-up lipid testing,[1] it is now generally preferable to follow non-HDL-C or ApoB levels over LDL-C when interpreting lipid results, particularly when triglyceride levels are ≥ 1.5 mmol/L. A recent survey conducted by the Canadian Association of Medical Biochemists and the Canadian Society of Clinical Chemistry indicates that patients across Canada can now present to laboratories nonfasting and receive a complete lipid profile.
Non-HDL-C or ApoB for predicting CVD risk
In population studies, non-HDL-C and ApoB can be considered as equivalent markers of total atherogenic lipoproteins and lipid-related CV risk and this applies to most individuals.[40] Publications since the 2016 update of these guidelines indicate a subgroup of individuals, estimated at between 8% and 23%, have discordance between ApoB and non-HDL-C levels in whom ApoB might be the better predictor of risk for coronary calcification[40] and ASCVD events.[41] Analysis of CV events in the large United Kingdom Biobank,[41] and metaanalysis of 110 prospective cohort registries of patients with or at risk for ASCVD,[42] however, showed an overall similar ability of non-HDL-C and ApoB to predict risk, but confirmed both of these measures to be superior to LDL-C. Recent consensus statements have concluded that non-HDL-C is currently a more practical choice because it incurs no additional expense to the patient or health care system.[43],[44] In Canada, the approach has been to allow clinicians to use either non-HDL-C or ApoB as their preferred parameter for assessment of risk and achievement of treatment targets, depending on their comfort level with the two measurements, availability of ApoB testing in their region, and when there might be a concern about discordance between the two measurements, as indicated previously. In the current guidelines, we are continuing this recommendation, while strongly urging the routine use of either non-HDL-C or ApoB instead of LDL-C as the lipid level of interest in initial lipid screening and as a treatment target in all patients with triglyceride level > 1.5 mmol/L.
Recommendation
- We recommend that for any patient with triglycerides > 1.5 mmol/L, non-HDL-C or ApoB be used instead of LDL-C as the preferred lipid parameter for screening (Strong Recommendation, HIgh-Quality Evidence).
PICO 2b: Is there evidence to support measurement of Lp(a) to improve risk stratification and dyslipidemia management in patients with and without previous CV events?
Lp(a) is an LDL-like particle in which ApoB is covalently bound to a plasminogen-like molecule called apolipoprotein (a).[45] Plasma concentrations of Lp(a) are not influenced by age, sex, fasting state, inflammation, or lifestyle factors, but are largely controlled by a single gene locus, LPA on chromosome 6, and are highly (> 90%) heritable.[46] Individual values are generally stable throughout life, thus, repeat measures are not required for risk assessment. Mendelian randomization studies have clearly shown that genetic variants in the LPA locus uniquely regulating Lp(a) levels are robustly associated with coronary heart disease risk, thereby strongly suggesting a causal association between Lp(a) and CVD.[47],[48] The risk of ASCVD increases with increasing Lp(a) levels 30 mg/dL in a dose-dependent fashion.[48]–[50] Among 7524 subjects in the Copenhagen Heart Study followed for 17 years, subjects with an Lp(a) concentration between 30 and 76 mg/dL had a 1.7-fold hazard ratio (HR) for MI and those with an Lp(a) level > 117 mg/dL had an adjusted HR of 2.7.[48] Among 6086 patients with a first MI and 6857 control participants from the INTERHEART study who were stratified according to ethnicity and adjusted for age and sex, Lp(a) concentrations > 50 mg/dL were associated with an increased risk of MI (odds ratio, 1.48; 95% confidence interval [CI], 1.43-1.67), independent of established CVD risk factors including diabetes mellitus, smoking, and high blood pressure.[51] Higher Lp(a) concentrations carried a particularly high population burden in South Asian and Latin American individuals.[51] An Lp(a) level > 50 mg/dL (> 100 nmol/L) is found in approximately 20% of individuals of European and South Asian descent, 40% of African American individuals, and fewer than 10% of East Asian individuals.[51],[52] Individuals with extreme elevations in Lp(a) have been shown to be at markedly high risk, with an event rate similar to that for other genetic dyslipidemias for which family screening is recommended (ie, heterozygous FH). As such, Lp(a) is a common but as yet not routinely measured ASCVD risk marker. Elevated Lp(a) level also increases the risk of recurrent ASCVD in a dose-dependent manner.[50],[53] Among 58,527 subjects from the Copenhagen General Population Study, 2527 subjects aged 20-79 years with a history of ASCVD and elevated Lp(a) were followed over a median of 5 years.[54] The adjusted major adverse CV events (MACE) incidence rate ratios were 1.28 (95% CI, 1.03-1.58) for subjects with an Lp(a) level of 10-49 mg/dL (18-104 nmol/L), 1.44 (95% CI, 1.12-1.85) for 50-99 mg/dL (105-213 nmol/L), and 2.14 (95% CI, 1.57-2.92) for those with Lp(a) ≥ 100 mg/dL (≥ 214 nmol/L).[54] In the randomized, controlled Further Cardiovascular Outcomes Research With PCSK9 Inhibition in Subjects With Elevated Risk (FOURIER) and Study to Evaluate the Effect of Alirocumab on the Occurrence of Cardiovascular Events in Patients Who Have Experienced an Acute Coronary Syndrome (ODYSSEY OUTCOMES) trials, high levels of Lp(a) were associated with an increased risk of recurrent CVD events in patients with established CVD irrespective of LDL cholesterol.[53],[54] Furthermore, alirocumab-associated reductions in Lp(a) reduced MACE in patients with a recent ACS independent of LDL-C.[54] Although these new data support the potential role of Lp (a) as a target of treatment in the future, there remains no evidence from RCTs that specifically lowering Lp(a) level leads to reductions in CV outcomes. It should also be noted that commonly used lipid-lowering therapies (ie, statins and ezetimibe) do not appreciably lower Lp(a) levels. The only available lipid-lowering therapies that lead to substantial lowering of Lp(a) include PCSK9 inhibitors, niacin, and apheresis, but relatively limited evidence exists for their use in patients with a high Lp(a) level. Newer investigational agents, such as antisense oligonucleotides and small interfering RNAs are currently being evaluated for CVD risk reduction in this patient population. Accordingly, Lp(a) is not currently considered a treatment target and repeat measures are therefore not indicated. Lp(a) testing is available across Canada, and is currently an insured laboratory test in most provinces, with the exception of Ontario and Manitoba.
Recommendation
- We recommend measuring Lp(a) level once in a person’s lifetime as a part of the initial lipid screening (Strong Recommendation; High-Quality Evidence).
- For all patients in the setting of primary prevention with a Lp(a) ≥ 50 mg/dL (or ≥ 100 nmol/L), we recommend earlier and more intensive health behaviour modification counselling and management of other ASCVD risk factors (Strong Recommendation; Expert Consensus).
Values and Preferences
There is a large body of evidence supporting the potential causal association between Lp(a) and future ASCVD.[50],[51],[55]–[58] The high prevalence of elevated Lp(a) level, the strength of association with incident and recurrent ASCVD events, and the potential to improve CV risk stratification, strongly justify universal screening to identify individuals with very high levels. Identification of high levels of Lp(a) is a useful consideration for shared decision-making in subjects across all ASCVD risk categories, but especially in younger patients, particularly those who have a very strong family history of premature ASCVD. Although further evidence that directly lowering Lp(a) level reduces ASCVD risk is pending, the finding of high Lp(a) should alert primary care practitioners to more actively pursue an overall ASCVD event risk assessment, including careful discussion of current health behaviours, consideration of age-appropriate vascular imaging studies for detecting early evidence of subclinical atherosclerosis in select individuals (eg, CAC score), and earlier introduction of statin or other lipid-lowering therapy, especially in intermediate-risk individuals and/or low-risk individuals with moderate elevations of LDL-C between 3.5 and 5 mmol/L. In the setting of secondary prevention, the presence of a high Lp(a) level is strongly predictive of recurrent events, and suggests the need for intensification of LDL-lowering therapy, including use of PCSK9 inhibitors. Furthermore, preliminary evidence suggests that treatment with PCSK9 inhibitors post ACS in patients with high Lp(a) reduces MACE independent of LDL-C lowering.[54] When clinicians are uncertain of the implications of elevated Lp(a), consultation with a lipid specialist might be considered.
PICO 3: In primary prevention, what is the evidence for CAC score to improve risk assessment? Specifically, should low CAC (or CAC = 0) score be used to avoid statin therapy in select individuals?
For primary prevention, most guidelines are on the basis of the concept of ASCVD risk assessment to help determine appropriateness and intensity of ASCVD risk factor modification. The primary prevention RCTs on which the recommendations are based, however, use clinical descriptors to identify patients eligible for study and, as a result, the patients eligible for the proven therapy. None of the algorithms available, including the FRS used in Canada, have been used to determine eligibility for any of the successful, primary prevention lipid-lowering trials. Even so, there is evidence to suggest that use of such algorithms is effective on a population level, more so than identification of patients on the basis of trial eligibility criteria.[59],[60] Despite this clinical utility, it has been repeatedly shown that typical ASCVD event risk algorithms can lead to substantial over or underestimation of ASCVD event risk,[61] and consequently, inappropriate risk factor management. Additionally, the value of these algorithms for predicting the presence and burden of atheroma is poor.[62],[63] Atheroma burden, the substrate that portends CV events, directly predicts ASCVD event risk in a graded fashion. This has been shown over decades with invasive angiography and more recently with coronary computed tomography, including noncontrast CAC scoring, the latter being highly applicable for assessment of patients who are asymptomatic, and possible candidates for primary prevention.[64],[65] Accordingly, the literature is replete with clinical studies reinforcing the concept that directly assessing the presence of atheroma, through CAC scoring, significantly improves the appropriate selection of patients who are likely to benefit from lipid modifying therapy.[66] Noncontrast CAC measurements are sensitive, reproducible, and can be performed rapidly with an average radiation dose of 0.89 mSv (compared with background annual radiation exposure of approximately 3.0 mSv). Evidence for improved C-statistic/net reclassification index after adjustment for standard risk factors (FRS) has been shown in multiple studies.[67],[69] The clinical decision-making utility of CAC measurements is best shown in middle-aged, intermediate-risk populations in whom the presence or absence of coronary artery calcification results in reclassification into higher or lower risk populations. A CAC measurement > 0 AU confirms the presence of atherosclerotic plaque. Increasing scores are directly proportional to increased ASCVD event risk.[69]–[72] A CAC measurement > 100 AU is associated with a high risk (> 2% annual risk) of an ASCVD event within 2-5 years and is generally an indication for intensive CV risk factor modification, including treatment of LDL-C. CAC > 300 AU places the patient in a very high risk category with a 10-year risk of MI/CV death of approximately 28%.[73] A CAC measurement of 0 AU, however, has a very high negative predictive value for ASCVD events in asymptomatic, low-risk adults within 2-5 years (negative predictive value, 95%-99%).[74] Importantly, although a CAC of 0 AU is indicative of a low event rate (1.5% per 10 years; 0.32-0.43 per 1000 person-years; 1.3-5.6 per 11.1 years),[70],[75]–[77] it is not indicative of a 0 event rate. This is likely because noncalcified soft plaque might be present; not all ASCVD events are mediated by vascular atheroma and atheromas might also progress in an unpredictable fashion. The variability in the development of clinical ASCVD with a CAC score of 0 AU is particularly evident in persons younger than 50 years of age, those with a strong family history of premature CVD events, or in the setting of severe CVD risk factors such as smoking, diabetes, poorly controlled hypertension, and in those with lifelong, genetic dyslipidemia (FH or elevated Lp[a]).[78]–[81] These are patient categories that in general would warrant aggressive ASCVD risk factor modification, even if CAC = 0 AU, to enhance the likelihood of maintaining as low an atheroma burden as possible over a lifetime. Conversely, if such high-risk patients do have CAC > 0 AU, this might provide a strong rationale for adherence to aggressive CVD risk factor modification,[82],[83] including lipid-lowering therapy or treatment intensification.[84],[85] The effects of statins on the progression of atherosclerosis cannot be assessed through serial CAC scores alone because it does not assess the status of noncalcific plaque. Therapy does not reduce and might even increase CAC scores despite regression of noncalcific plaque components.[86] Accordingly, repeat CAC scanning is not recommended unless risk factor modification has been deferred through patient-physician shared decision-making. Although CAC provides direct evidence of atherosclerotic plaque and a quantitative assessment of risk of attendant ASCVD events, controversy exists because of a paucity of large placebo-controlled RCTs and its cost-effectiveness for identification of patients suitable for statin therapy is uncertain,[87] even when applied only to the intermediate-risk group identified using risk algorithms. Importantly, at present, CAC scoring is not uniformly available or uniformly funded in Canada, and there are no cost-effectiveness analyses that represent the Canadian context.
Recommendation
- We suggest that CAC screening using computed tomography imaging might be considered for asymptomatic adults 40 years of age or older and at intermediate risk (FRS 10%-20%) for whom treatment decisions are uncertain (Strong Recommendation; Moderate-Quality Evidence).
- We recommend that CAC screening using computed tomography imaging not be undertaken for: (1) high-risk individuals; (2) patients receiving statin treatment; or (3) most asymptomatic, low-risk adults (Strong Recommendation; Moderate-Quality Evidence).
Recommendation
- We suggest that CAC screening might be considered for a subset of low-risk individuals 40 years of age or older with a family history of premature ASCVD (men 55 years or younger; women 65 years or younger) in addition to identifying known genetic causes of ASCVD such as elevated Lp(a) level or FH (Weak Recommendation; Low-Quality Evidence).
Values and Preferences
Patients with modifiable ASCVD risk factors should be counselled with respect to the potential merit of preventing atherosclerosis itself, the substrate for clinical ASCVD events in the long term, through comprehensive ASCVD risk factor management. As outlined elsewhere, RCTs show the ASCVD risk reduction value of statin therapy in patients with intermediate risk and additional ASCVD risk factors (eg, HOPE 3,[16] Justification for the Use of Statins in Prevention: An Intervention Trial Evaluating Rosuvastatin [JUPITER][88]) in the absence of CAC testing or any testing to identify preclinical atherosclerosis. Accordingly, the patient-physician decision often does not require CAC scoring but might be strongly influenced by these other factors, including family history of premature ASCVD, other features suggesting genetic causes of dyslipidemia, or side effects of statin therapy. In some low to intermediate-risk subjects, it might be reasonable to withhold statin therapy for CAC = 0 AU because of a favourable intermediate-term outcome. Exceptions would include cigarette smokers, patients with diabetes, those with poorly controlled hypertension, genetic dyslipidemias such as FH or elevated Lp(a) level, and patients with strong family history of premature ASCVD events. If available, a CAC > 100 AU is an indication for statin therapy regardless of FRS. For those with a CAC of 1-99 AU, individual decision-making is required because risk will not be reclassified and would remain intermediate. If a decision is made to withhold statin or lipid-modifying therapy on the basis of CAC = 0, this decision should be reevaluated during follow-up or if clinical circumstances change. CAC scoring should rarely be performed sooner than within 5 years to aid in this reevaluation. Finally, this section is restricted to 1138 Canadian Journal of Cardiology Volume 37 2021 application in patients who are at least 40 years of age for whom the traditional FRS assessment applies. Prevalence of calcification is a sequential aspect of the atherosclerotic process and might be absent in the early phases. Although CAC has been studied extensively for ASCVD risk prediction, the prevalence of CAC is lower in young patients compared with middle-aged and older patients and also in women vs men younger than 50 years of age.
PICO 4: In secondary prevention, what is the most appropriate lipid/lipoprotein threshold for the intensification of therapy?
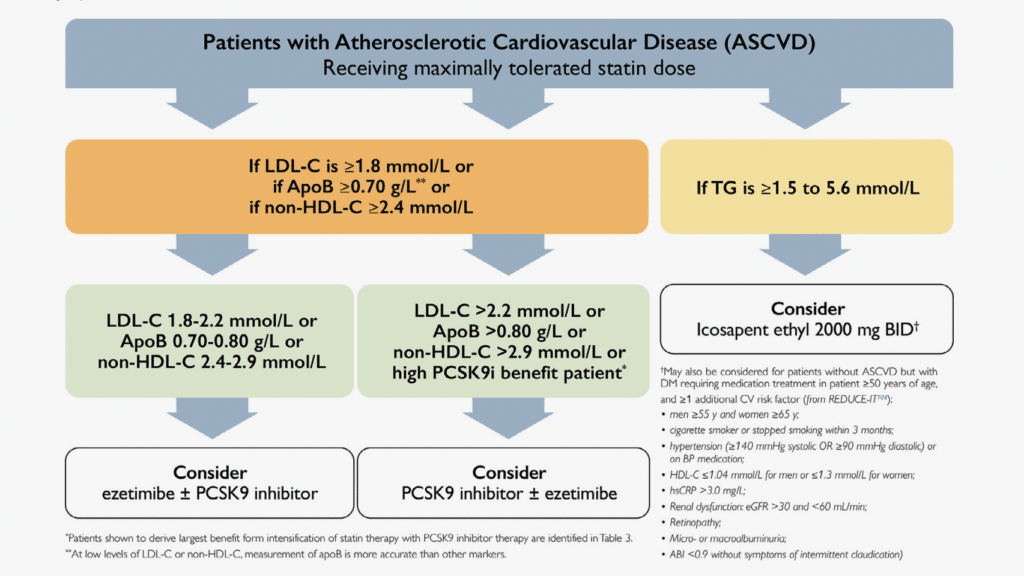
The totality of evidence from observational, pathophysiological, epidemiological, and Mendelian randomization studies and RCTs of lipid-lowering therapies indicate a causal relationship between LDL-C (as well as non-HDL-C and ApoB) and ASCVD and show that lower concentrations of plasma LDL-C levels are associated with a lower risk of ASCVD events extending to very low LDL-C concentrations (< 0.5 mmol/L).[15],[89]–[96] In RCTs, however, the absolute benefits of therapy were higher in subsets of patients with higher pretreatment LDL-C and/or additional ASCVD event risk enhancers who were at higher absolute risk. To date, no clear target to which LDL-C or non HDL-C or ApoB levels should be lowered is clearly identified in RCTs, because such trials have generally used thresholds of LDL-C (or non-HDL-C or ApoB) levels for initiation or intensification of lipid-lowering therapies and fixed-dose lipid-lowering drugs (this pertains to statin RCTs and to RCTs that have used the additional use of nonstatin lipid-lowering agents, such as ezetimibe and PCSK9 inhibitors). Exceptions are the Scandinavian Simvastatin Survival Study (4S) trial in which the statin dose was up- or down-titrated aiming for within-trial total cholesterol levels of 3.0-5.2 mmol/L,[97] the Improved Reduction of Outcomes: Vytorin Efficacy International Trial (IMPROVE-IT), which allowed for up-titration of simvastatin to 80 mg daily for in-trial LDL-C levels > 2.0 mmol/L,[98] and the ODYSSEY OUTCOMES trial in patients with a recent ACS, which allowed up-and down-titration of alirocumab aiming for an LDL-C target of 0.65- 1.3 mmol/L; however, in these trials no randomized comparison with alternate lipid targets was performed.[90] Additionally, a number of trials comparing different intensities of statin treatment (lower vs higher statin dose) in secondary ASCVD prevention showed benefits for more intensive statin therapy; however, these trials did not explore targets of LDL-C lowering.[99],[100] One RCT conducted in patients with a recent ischemic stroke showed reductions in major ASCVD events in patients allocated to a strategy of lower LDL-C (< 1.8 mmol/ L) vs higher targets (2.3-2.8 mmol/L).[101] Nevertheless, the lower LDL-C target in this trial is similar to the threshold for intensification of lipid-lowering therapy used in other recent trials and recommended in this guideline document.[89],[102] A number of studies have shown improved ASCVD outcomes in secondary prevention patients reaching lower in-trial LDLC levels, but these trials are observational and did not test targets of therapy.[103],[104] Therefore, we recommend the use of thresholds for intensification of lipid therapy in secondary prevention. Most recent large RCTs have used an LDL-C threshold of 1.8 mmol/L for intensification of lipid-lowering therapy with nonstatin drugs in secondary ASCVD prevention patients receiving a maximally tolerated statin dose. Using this threshold, it is expected that most patients will achieve low and very low LDL-C levels, similar to those reached in clinical trials.[90],[91] The IMPROVE-IT trial showed benefit of ezetimibe when used in addition to statin therapy in patients with a recent ACS.[98] The threshold for the additional use of ezetimibe was an LDL-C of 1.3 mmol/L, although in IMPROVE-IT most patients had a higher baseline LDL-C (average 2.45 mmol/L), statin therapy was restricted to only simvastatin (more potent statins were not used) and the modest 6% relative risk reduction was attained only after a long period of treatment (median 6 years). Therefore, we recommend the more robust LDL-C threshold of ≥ 1.8 mmol/L (or percentile equivalent non-HDL-C of ≥ 2.4 mmol/L or ApoB of ≥ 0.7 g/L). Recent analyses of the large PCSK9 inhibitor trials (FOURIER89 and ODYSSEY OUTCOMES[90]) have identified subsets of patients with established CVD who are at very high risk and who derived the largest absolute benefit for intensification of lipid-lowering therapy with evolocumab and alirocumab, respectively. This includes patients with recent ACS and those with ASCVD and additional CV risk enhancers including diabetes mellitus, metabolic syndrome, polyvascular disease (vascular disease in ≥ 2 arterial beds), symptomatic peripheral artery disease, history of MI, MI in the past 2 years, previous coronary artery bypass graft surgery, LDL ≥ 2.6 mmol/L, heterozygous FH and Lp(a) ≥ 60 mg/dL.[90],[105]–[113] Intensification of lipid-lowering therapy with PCSK9 inhibitors is especially recommended in these subsets of very high risk patients (see Table 3), with or without the additional use of ezetimibe, which was used in only a small number of patients in these trials. Use of PCSK9 inhibitor therapy in these subsets of patients was shown to result in rapid and large reductions in LDL-C and in significant CVD event reduction. In most other secondary prevention patients, the use of ezetimibe followed by PCSK9 inhibitor therapy is recommended when the LDL-C ≥ 1.8 mmol/L. The previous 2016 CCS dyslipidemia guidelines did not emphasize the role of plasma triglyceride levels as a threshold or target for lipid-lowering therapy aimed at reducing CVD risk.[1] However, the recent Reduction of Cardiovascular Events With Icosapent Ethyl-Intervention Trial (REDUCE-IT) showed a CV risk reduction (including reduction in CV death) in patients with ASCVD (as well as in those 50 years old or older with type 2 diabetes requiring medication treatment and at least 1 additional CVD risk factor) receiving moderate and high-intensity statin therapy with triglyceride levels of 1.5-5.6 mmol/L and LDL-C levels of 1.1- 2.6 mmol/L.[114]
Recommendation
- We recommend use of high-intensity statin therapy in addition to appropriate health behaviour modifications for all secondary prevention CVD patients. For patients who do not tolerate a high-intensity statins, Pearson et al. 1139 CCS Dyslipidemia Guidelines we recommend the maximally tolerated statin dose (Strong Recommendation; High-Quality Evidence).
- We recommend intensification of lipid-lowering therapy with a PCSK9 inhibitor (evolocumab or alirocumab)—with or without the additional use of ezetimibe—for secondary CV prevention patients shown to derive the largest benefit from PCSK9 inhibitor therapy in whom LDL-C remains ≥ 1.8 mmol/L (or non-HDL-C ≥ 2.4 mmol/L or ApoB ≥ 0.7 g/L) while receiving the maximally tolerated statin dose (Fig. 3; Strong Recommendation; Moderate-Quality Evidence). Secondary prevention patients shown to derive the largest benefit from intensification of statin therapy with PCSK9 inhibitor therapy are defined in Table 3.
- We recommend intensification of lipid-lowering therapy with ezetimibe and/or PCSK9 inhibitor therapy for all secondary prevention CVD patients in whom LDL-C remains ≥ 1.8 mmol/L (or non-HDL-C ≥ 2.4 mmol/L or ApoB ≥ 0.7 g/L) while receiving the maximally tolerated statin dose. (Strong Recommendation; High-Quality Evidence). If ezetimibe is used initially and LDL-C remains ≥ 1.8 mmol/L (or nonHDL-C ≥ 2.4 mmol/L or ApoB ≥ 0.7 g/L) PCSK9 inhibitor therapy is recommended (Strong Recommendation; High-Quality Evidence). It should be noted that one recommendation on the basis of the evidence review of PICO question 4 were overlapping with a recommendation for PICO question 5 and appear as part of that later section (Recommendation 15).
Values and Preferences
On the basis of strong evidence for the benefit of intensive LDL-C lowering in secondary prevention, additional lipid-lowering therapy with ezetimibe and PCSK9 inhibitors might also be considered for ASCVD patients with an LDL-C < 1.8 mmol/L, especially for patients considered to be at high risk for recurrent ASCVD events. When initiating intensified lipid-lowering therapy with nonstatin drugs, cost, and access to such therapies should be considered. There is no evidence to suggest any CV or other risks associated with low and very low LDL-C levels in trials with moderate duration of follow-up.[104],[115],[116] Therefore, if intensified lipid-lowering therapy initiated for the previously listed thresholds result in low and very low LDL-C levels, lipid-lowering therapy does generally not require down-titration dose adjustment.
Practical Tip
Although there is very good evidence supporting the use of PCSK9 inhibitors in patients with ASCVD (especially those listed in Table 3), access might be limited by provincial drug plan coverage in many jurisdictions. Patients with or without private drug plan coverage might need to pay some portion of the cost of these expensive medications. Patient support programs for these medications could be investigated to assist. Clinicians should discuss the indication and potential benefits of a PCSK9 inhibitor with the patient, along with the coverage issues and the potential costs to them. Shared decision-making remains key.

PICO 5: In adults already receiving (or intolerant to) statins, what is the role of nonstatin drugs to reduce CVD risk?
Ezetimibe. Ezetimibe is a cholesterol absorption inhibitor that lowers LDL-C by approximately 20% in addition to a statin regimen or up to 15% as monotherapy. Only in 1 double-blind, RCT has the efficacy of ezetimibe been assessed in reducing CV risk. The IMPROVE-IT showed that ezetimibe 10 mg daily, compared with placebo and used in addition to statin therapy, showed a modest reduction in CV events in 18,144 patients with an ACS within the preceding 10 days.[98] The primary composite outcome of death from CV causes, major coronary events, and nonfatal stroke was 2% lower with ezetimibe (32.7 vs 34.7%; HR, 0.94; 95% CI, 0.89- 0.99) for a number need to treat of 50 over 7 years. There were no significant differences between groups in the prespecified safety end points. This evidence informed the 2016 guideline recommendation for ezetimibe as second-line therapy to reduce CV risk in patients with ASCVD if their LDLC targets were not reached with maximally tolerated statin therapy.[1] Subsequently, in the Heart Institute of Japan-Proper Level of Lipid Lowering With Pitavastatin and Ezetimibe in Acute Coronary Syndrome (HIJ-PROPER) trial open-label pitavastatin with ezetimibe (target LDL-C < 1.8 mmol/L) was compared with pitavastatin monotherapy (target LDL-C 2.3-2.6 mmol/L) in 1734 Japanese patients with an ACS. Over 3.9 years, the primary composite outcome of all-cause death, nonfatal MI, nonfatal stroke, unstable angina, and ischemia-driven revascularization was not significantly different between groups (32.8 vs 36.9%; HR 0.89; 95% CI, 0.76-1.04).[117]
PCSK9 inhibitors. Inhibitors of PCSK9 are recently available monoclonal antibodies that lower LDL-C between 50% and 70% when used in addition to statin therapy or as monotherapy.[118] Currently, two PCSK9 inhibitors are approved for use in Canada: alirocumab and evolocumab. Both are approved for the treatment of FH or ASCVD in patients as an adjunct to diet and maximally tolerated statin therapy (with or without ezetimibe) who require additional lowering of LDL-C. The FOURIER trial enrolled 27,564 patients with clinical ASCVD and additional CVD risk factors whose LDL-C remained ≥ 1.8 mmol/L despite maximally tolerated statin therapy. Patients were randomized to receive evolocumab (140 mg subcutaneously (SC) every 2 weeks or 420 mg SC monthly) or placebo.[89] Baseline LDL-C was 2.4 mmol/L, which after 48 weeks was reduced to a median of 0.8 mmol/L (interquartile range, 0.5-1.2 mmol/L) in the evolocumab group. After 2.2 years of follow-up, the primary outcome of CV death, nonfatal MI, nonfatal stroke, hospitalization for unstable angina, and coronary revascularization was lower with evolocumab (9.8% vs 11.3%; HR, 0.85; 95% CI, 0.79- 0.92) for a number needed to treat of 67. Evolocumab also reduced the secondary end point of CV death, nonfatal MI, and nonfatal stroke (5.9% vs 7.4%; HR, 0.80; 95% CI, 0.73- 0.88). There was no significant difference in CV or all-cause death. Serious adverse events were similar between groups, although injection site reactions were higher with evolocumab (2.1% vs 1.6%; P < 0.001). In the ODYSSEY OUTCOMES trial alirocumab was evaluated in 18,924 patients with a recent (1-12 months) ACS whose LDL-C was ≥ 1.8 mmol/L despite maximally tolerated statin therapy.[90] Participants were randomized to alirocumab (75 mg SC every 2 weeks to achieve an LDL-C of 0.6-1.3 mmol/L) or placebo. The dose of alirocumab was increased to 150 mg SC every 2 weeks if a participant’s LDL-C level remained > 1.3 mmol/L or decreased or discontinued if their LDL-C level was < 0.6 mmol/L. The primary outcome of death from coronary heart disease, nonfatal MI, fatal or nonfatal ischemic stroke, or unstable angina requiring hospitalization was lower with alirocumab (9.5% vs 11.1%; HR 0.85; 95% CI, 0.78-0.93) for a number needed to treat of 63 over 2 years. All-cause mortality was numerically lower with alirocumab (3.5% vs 4.1%), but on the basis of the authors’ prespecified hierarchical testing, it is debatable whether this can be considered statistically significant. There was no significant difference in CV death between groups. There was no significant difference in serious adverse events, but injection site reactions were more common with alirocumab (3.8% vs 2.1%; P < 0.001). A recent meta-analysis of 23 trials (including FOURIER and ODYSSEY OUTCOMES) compared PCSK9 inhibitors with control in 60,723 patients.[119] There was a significant reduction in MACE (6.2% vs 8.2%; risk ratio, 0.83; 95% CI, 0.78-0.88) with no significant difference in all-cause mortality (risk ratio 0.93; 95% CI, 0.85-1.02) or safety outcomes. Of note, these trials had short follow-up (median of 2.8 years) and therefore might not have been of sufficient duration to observe a mortality benefit. Although ezetimibe or a PCSK9 inhibitor are reasonable options as monotherapy in patients with complete statin intolerance for LDL-C lowering, there is limited evidence to support either class as an alternative to statin therapy for ASCVD risk reduction. The Study of Alirocumab (REGN727/SAR236553) in Patients With Primary Hypercholesterolemia and Moderate, High, or Very High Cardiovascular (CV) Risk, Who Are Intolerant to Statins (ODYSSEY ALTERNATIVE) trial enrolled 314 patients with statin intolerance who were randomized to alirocumab 75 mg SC every 2 weeks, ezetimibe 10 mg daily, or atorvastatin 20 mg daily.[120] At 24 weeks, alirocumab reduced LDL-C by a mean difference of 30% compared with ezetimibe. Skeletal muscle-related adverse effects were high overall, but significantly lower with alirocumab (33%) vs atorvastatin (46%) and similar to ezetimibe (41%). The Goal Achievement After Utilizing an Anti-PCSK9 Antibody in Statin Intolerant Subjects3 (GAUSS-3) trial included 218 patients considered to have previous statin intolerance who were randomized to evolocumab 420 mg SC monthly or ezetimibe 10 mg orally daily.[121] Evolocumab showed a significantly greater reduction in LDL-C compared with ezetimibe (mean difference, 36%) at 24 weeks. The incidence of muscle symptoms was relatively high in both groups, but the difference was not statistically significant (21% vs 29%; P = 0.17). Clinical trials have shown that PCSK9 inhibitors are effective at lowering LDL-C in patients with heterozygous FH[102],[120] and in certain patients with homozygous FH,[91] but there is currently a paucity of ASCVD outcome data in these populations.

Primary prevention
There are currently no RCT data supporting the use of PCSK9 inhibitors to reduce CV events in patients who do not have established ASCVD (ie, primary CV prevention) or FH.
IPE. Until recently, contemporary trials of omega-3 fatty acid supplements have not shown a CV benefit in patients with or without CVD.[123],[124] Previously, the Japan EPA Lipid Intervention Study (JELIS) showed a reduction in CV events with 1800 mg daily of eicosapentaenoic acid (EPA) combined with a statin, compared with statin monotherapy, in Japanese patients with a total cholesterol ≥ 6.5 mmol/L; however, it was an open-label trial and the primary outcome was driven by a minor reduction in unstable angina.[125] The REDUCE-IT assessed the effect of a pharmaceutical formulation of purified ethyl EPA (IPE), which was recently approved by Health Canada.[114] In total, 8179 patients were included with established ASCVD (or diabetes and ≥ 1 ASCVD risk factor) who were receiving statin therapy but had an elevated fasting triglyceride level of 1.5-5.6 mmol/L (baseline 2.4 mmol/L). Most patients (71%) were in the secondary prevention cohort. Participants were randomized to 2000 mg of IPE orally twice daily (4 g total per day) or mineral oil as placebo. At 1 year, participants’ triglyceride level in the IPE group was modestly reduced by 0.4 mmol/L (approximately 18%) from baseline. IPE reduced the primary outcome of CV death, nonfatal MI, nonfatal stroke, unstable angina, or CV revascularization (17.2% vs 22.0%; HR, 0.75; 95% CI, 0.68-0.83) for a number needed to treat of 21 over 4.9 years. IPE also significantly reduced the composite of CV death, nonfatal MI, and nonfatal stroke (11.2% vs 14.8%; HR, 0.74; 95% CI, 0.65-0.83), as well as CV death (4.3% vs 5.2%; HR, 0.80; 95% CI, 0.66-0.98), but not all-cause death. Atrial fibrillation and peripheral edema were significantly higher with IPE. Because IPE is a purified form of ethyl EPA, the results of REDUCE-IT cannot be extrapolated to other nonprescription omega-3 fatty acids, which typically contain a mixture of EPA and docosahexaenoic acid (DHA). The Outcomes Study to Assess Statin Residual Risk Reduction With Epanova in High CV Risk Patients With Hypertriglyceridemia (STRENGTH) trial aimed to evaluate a pharmaceutical carboxylic acid formulation of EPA and DHA (referred to as omega-3 CA) to prevent MACE in 13,078 patients with hypertriglyceridemia (2.0-5.6 mmol/L), low HDL-C (< 1.2 mmol/L for women and < 1.1 mmol/L for men) who were receiving statin therapy, and were at increased risk of CVD.[128] Patients were randomized to receive 4 g/d of omega-3 CA or corn oil placebo. The trial was discontinued prematurely after a median follow-up of 3.5 years for futility. The primary end point of CV death, nonfatal MI, nonfatal stroke, unstable angina requiring hospitalization, and coronary revascularization was not significantly different between groups (12.0% vs 12.2%; HR, 0.99; 95% CI, 0.90-1.09). Patient-reported gastrointestinal disorders were more common in patients in the omega-3 CA group (24.7% vs 14.7%).
Other therapies. There are no new recommendations regarding the use of fibrates, niacin, and bile acid sequestrants since the 2016 guidelines.[1]
Ongoing trials
There are a number of ongoing trials of nonstatin therapy. Effect of Evolocumab in Patients at High Cardiovascular Risk Without Prior Myocardial Infarction or Stroke (VESALIUSCV) is designed to examine the effect of evolocumab at reducing MACE in patients without a previous MI or stroke but who are at high risk of CVD.[127] Inclisiran is an experimental small interfering RNA molecule that inhibits the translation of PCSK9. In the phase III Trial to Evaluate the Effect of Inclisiran Treatment on Low Density Lipoprotein Cholesterol in Subjects With Heterozygous Familial Hypercholesterolemia (ORION-9), Inclisiran for Participants With Atherosclerotic Cardiovascular Disease and Elevated Low-density Lipoprotein Cholesterol (ORION-10), and Inclisiran for Subjects With ASCVD or ASCVD-Risk Equivalents and Elevated Low-density Lipoprotein Cholesterol (ORION-11) trials, inclisiran showed LDL-C lowering in patients with heterozygous FH or with, or at high risk of, atherosclerotic CVD.[128],[129] The ongoing phase III A Randomized Trial Assessing the Effects of Inclisiran on Clinical Outcomes Among People With Cardiovascular Disease (ORION-4) is evaluating whether this LDL-C reduction with inclisiran translates to a reduction in MACE among patients with CVD.[130] The Effect of Dalcetrapib vs Placebo on CV Risk in a Genetically Defined Population With a Recent ACS (dalGenE) study aims to assess the effect of dalcetrapib, a cholesteryl ester transfer protein inhibitor (not approved by Health Canada), in patients with a recent ACS and specific genotype.[131] The Cholesterol Lowering via Bempedoic Acid, an ACL-Inhibiting Regimen (CLEAR) Outcomes trial is evaluating the effect of bempedoic acid, a novel adenosine triphosphate (ATP) citrate lyase inhibitor not approved in Canada, in patients with, or at high risk for, ASCVD who are statin-intolerant.[132] Finally, the Pemafibrate to Reduce Cardiovascular Outcomes by Reducing Triglycerides in Patients With Diabetes (PROMINENT) trial is determining whether pema-fibrate, a peroxisome proliferator-activated receptor alpha agonist (which is not approved for use in Canada) reduces MACE in patients with type 2 diabetes mellitus, elevated triglycerides, and low HDL-C.[133]
- We recommend the use of IPE to decrease the risk of CV events in patients with ASCVD, or with diabetes and ≥ 1 CVD risk factors, who have an elevated fasting triglyceride level of 1.5-5.6 mmol/L despite treatment with maximally tolerated statin therapy (Strong Recommendation; High-Quality Evidence). Refer to Figure 3.
- We recommend the use of a PCSK9 inhibitor (alirocumab or evolocumab) to lower LDL-C level in patients with heterozygous FH without clinical ASCVD whose LDL-C remains above the target (ie, LDL-C ≥ 2.5 mmol/L or < 50% reduction from baseline; or ApoB ≥ 0.85 mg/dL or non-HDL-C ≥ 3.2 mmol/L) despite maximally tolerated statin therapy with or without ezetimibe therapy (Strong Recommendation; High-Quality Evidence).
- We recommend the use of a PCSK9 inhibitor (alirocumab or evolocumab) for patients with heterozygous FH and ASCVD whose LDL-C remains above the threshold ≥ 1.8 mmol/L (or ApoB ≥ 0.7 mg/dL or non-HDL-C ≥ 2.4 mmol/L) despite maximally tolerated statin therapy, with or without ezetimibe (Strong Recommendation; High-Quality Evidence). It should be noted that 2 recommendations on the basis of the evidence review of PICO question 5 were overlapping with recommendations made for PICO question 4 and appear as part of that earlier section (Recommendations 1 and 2).
Values and Preferences
None of these agents have been evaluated in RCTs against each other. Therefore, it is difficult to assess the relative benefit of each therapy. Also, to date these agents have primarily been evaluated in patients with preexisting ASCVD (ie, secondary prevention). The choice of agent should be on the basis of individual patient factors, their values and preferences, and practical considerations, such as access, cost, and adherence. Because ezetimibe lowers LDL-C level by approximately 20% when used in addition to a statin, if a patient’s LDL-C is well above the threshold for therapy intensification (ie, > 2.2 mmol/L or > 20% above threshold), it might be preferable to consider a PCSK9 inhibitor as second-line therapy. However, because of cost considerations, some insurance providers might require a trial of ezetimibe before approving the use of a PCSK9 inhibitor. IPE should be preferentially reserved for patients aged ≥ 45 years of age (or ≥ 50 years of age with ≥ 1 CVD risk factor) who are receiving maximally tolerated statin therapy but have a residual elevated triglyceride level (1.5-5.6 mmol/L). Because IPE is a purified form of ethyl EPA, it should not be inferred that the same CV benefits could be derived from the consumption of omega-3 polyunsaturated fatty acid (PUFA) formulations that include EPA alone, EPA and DHA mixtures, or fish oils from supplements or dietary sources. The recommendation for treatment of patients with FH is on the basis of the 2018 update to the CCS position statement on FH.[134] The recommendation for PCSK9 inhibitors to lower LDL-C level is on the basis of highquality evidence; however, there is a relative paucity of RCT evidence to support any agent to reduce the risk of CV events in FH patients.
Practical Tip
Unlike the use of PCSK9 inhibitors in patients with ASCVD, access to these medications is covered by most provincial drug plans for patients with heterozygous FH (with or without ASCVD) with LDL-C level above the threshold. Although the evidence for IPE to decrease the risk of CV events in patients with ASCVD, or with diabetes and ≥ 1 CVD risk factors is good, it is relatively new and most provincial drug plans do not yet cover this expensive medication. Private plans might cover this drug for patients on the basis of specific criteria and there is a manufacturer patient assistance program that might facilitate access. As part of shared decision-making, clinicians should discuss the indication and potential benefits of IPE, as well as the coverage issues and the potential patient costs.
PICO 6: In primary and secondary prevention, what is the evidence for CV benefit of omega-3 from (1) dietary sources; and/or (2) over-the-counter formulations/supplements?
Despite the success of the REDUCE-IT trial in showing a purified prescription IPE at 4 g/d reduces major CVD events in statin-treated patients with elevated triglyceride levels who have established CVD or diabetes and at least 1 CVD risk factor,[114] supplementation with over-the-counter long-chain omega-3 PUFAs marketed as natural health products in Canada that include EPA alone, EPA and DHA mixtures, or fish oils from supplements or dietary sources does not offer any clear advantages for CVD event risk reduction. We updated a systematic review and meta-analysis of RCTs with data from 2 subsequently completed RCTs, A Study of Cardiovascular Events in Diabetes (ASCEND)[135] and Vitamin D and Omega-3 Trial (VITAL),[136] which failed to show a clear CV benefit of supplementation with long chain omega-3 PUFAs in more than 130,000 randomized participants.[137] Another large CVD outcomes trial of a pharmaceutical drug of mixed long-chain omega-3 (largely EPA and DHA) carboxylic acids (omega-3 CA) at 4 g/d with similar entry criteria to the REDUCE-IT trial was also discontinued early by the data safety monitoring board for futility with the drug unlikely to show a benefit to patients.[126] Pooled evidence from RCTs[138]–[140] and individual large RCTs,[141] however, have shown consistent triglyceride-lowering effects at high doses (2-4 g/d) of omega-3 PUFAs, independent of CVD event risk reduction.
Recommendation
- We do not recommend the use of over-the-counter omega-3 polyunsaturated fatty acid supplements (marketed as natural health products in Canada) to reduce CVD risk (Strong Recommendation; HighQuality Evidence).
Values and Preferences
Although there is no apparent overall CVD event risk benefit, patients might choose to use these supplements for other indications including the management of high triglycerides, for which very high doses are required (4 g/d), and for which fibrates are generally more effective. Individuals should be aware that, in addition to marine sources, there are different preparations of long-chain omega-3 PUFAs high in DHA and EPA from algal and yeast sources, both of which are suitable for vegans. There is also alpha-linolenic acid from plant sources that do not contain DHA or EPA including flax seeds, chia seeds, and some oils such as canola and soybean oil, which have little or no effect on triglycerides.
References
- Bellamy L, Casas JP, Hingorani AD, Williams DJ. Pre-eclampsia and risk of cardiovascular disease and cancer later in life: a systematic review and meta-analysis. BMJ 2007;335:974.
- Grandi SM, Filion KB, Yoon S, et al. Cardiovascular disease-related morbidity and mortality in women with a history of pregnancy complications. Circulation 2019;139:1069–79.
- Auger N, Fraser WD, Schnitzer M, et al. Recurrent pre-eclampsia and subsequent cardiovascular risk. Heart 2017;103:235–43.
- Ray JG, Vermeulen MJ, Schull MJ, Redelmeier DA. Cardiovascular Health After Maternal Placental Syndrome (CHAMPS): population-based retrospective cohort study. Lancet 2005;366:1797–803.
- Al-Nasiry S, Ghossein-Doha C, Polman SEJ, et al. Metabolic syndrome after pregnancies complicated by pre-eclampsia or small for gestational age: a retrospective cohort. BJOG 2015;122:1818–23.
- Grand’Maison S, Pilote L, Landry T, Okano M, Dayan N. Markers of vascular dysfunction after hypertensive disorders of pregnancy: a systematic review and meta-analysis. Hypertension 2016;68:1447–58.
- Dayan N, Schlosser K, Stewart DJ, et al. Circulating microRNAs implicate multiple atherogenic abnormalities in the long-term cardiovascular sequelae of preeclampsia. Am J Hypertens 2018;31:1093–7.
- Arnett DK, Blumenthal RS, Albert MA, et al. 2019 ACC/AHA guideline on the primary prevention of cardiovascular disease. A report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation 2019;140:e596–646.
- Gamble DT, Brikinns B, Myint PK, Bhattacharya S. Hypertensive disorders of pregnancy and subsequent cardiovascular disease: current national and international guidelines and the need for future research. Front Cardiovasc Med 2019;6:55.
- Smith GN, Pudwell J, Roddy M. The maternal health clinic: a new window of opportunity for early heart disease screening and intervention for women with pregnancy complications. J Obstet Gynaecol Can 2013;35:831–9.
- Bateman BT, Hernandez-Diaz S, Fischer MA, et al. Statins and congenital malformations: cohort study. BMJ 2015;350:h1035.
- Pollack PS, Shields KE, Burnett DM, et al. Pregnancy outcomes after maternal exposure to simvastatin and lovastatin. Birth Defects Res A Clin Mol Teratol 2005;73:888–96.
- Edison RJ, Muenke M. Central nervous system and limb anomalies in case reports of first-trimester statin exposure. N Engl J Med 2004;350:1579–82.
- Anderson TJ, Gregoire J, Hegele RA, et al. 2012 update of the Canadian Cardiovascular Society guidelines for the diagnosis and treatment of dyslipidemia for the prevention of cardiovascular disease in the adult Can J Cardiol 2013;29:151–67.
- Carmena R, Duriez P, Fruchart JC. Atherogenic lipoprotein particles in atherosclerosis. Circulation 2004;109(23 suppl 1). III2-7.
- Kathiresan S, Otvos JD, Sullivan LM, et al. Increased small low-density lipoprotein particle number: a prominent feature of the metabolic syndrome in the Framingham Heart Study. Circulation 2006;113:20–9.
- Sniderman AD, Williams K, Contois JH, et al. A meta-analysis of low-density lipoprotein cholesterol, non-high-density lipoprotein cholesterol, and apolipoprotein B as markers of cardiovascular risk. Circ Cardiovasc Qual Outcomes 2011;4:337–45.
- Boekholdt SM, Arsenault BJ, Mora S, et al. Association of LDL cholesterol, non-HDL cholesterol, and apolipoprotein B levels with risk of cardiovascular events among patients treated with statins: a meta-analysis. JAMA 2012;307:1302–9.
- Langsted A, Freiberg JJ, Nordestgaard BG. Fasting and nonfasting lipid levels: influence of normal food intake on lipids, lipoproteins, apolipo proteins, and cardiovascular risk prediction. Circulation 2008;118:2047–56.
- Sidhu D, Naugler C. Fasting time and lipid levels in a community-based population: a cross-sectional study. Arch Intern Med 2012;172:1707–10.
- Wilkins JT, Li RC, Sniderman A, Chan C, DM Lloyd-Jones. Discordance between apolipoprotein B and LDL-cholesterol in young adults predicts coronary artery calcification: the CARDIA study. J Am Coll Cardiol 2016;67:193–201.
- Welsh C, Celis-Morales CA, Brown R, et al. Comparison of conventional lipoprotein tests and apolipoproteins in the prediction of cardiovascular disease. Circulation 2019;140:542–52.
- Perera R, McFadden E, McLellan J, et al. Optimal strategies for monitoring lipid levels in patients at risk or with cardiovascular disease: a systematic review with statistical and cost-effectiveness modelling. Health Technol Assess 2015;19:1–401. vii-viii.
- Langlois MR, Chapman MJ, Cobbaert C, et al. Quantifying atherogenic lipoproteins: current and future challenges in the era of personalized medicine and very low concentrations of LDL cholesterol. A consensus statement from EAS and EFLM. Clin Chem 2018;64:1006–33.
- Nordestgaard BG, Langlois MR, Langsted A, et al. Quantifying atherogenic lipoproteins for lipid-lowering strategies: consensus-based recommendations from EAS and EFLM. Atherosclerosis 2020;294:46–61.
- Witztum JL, Ginsberg HN. Lipoprotein (a): coming of age at last. JLipid Res 2016;57:336–9.
- Langsted A, Kamstrup PR, Nordestgaard BG. Lipoprotein(a): fasting and nonfasting levels, inflammation, and cardiovascular risk. Athero-sclerosis 2014;234:95–101.
- Clarke R, Peden JF, Hopewell JC, et al. Genetic variants associated with Lp(a)lipoprotein level and coronary disease. N Engl J Med 2009;361:2518–28.
- Kamstrup PR, Tybjaerg-Hansen A, Steffensen R, Nordestgaard BG. Genetically elevated lipoprotein(a) and increased risk of myocardial infarction. JAMA 2009;301:2331–9.
- Emerging Risk Factors CollaborationErqou S, Kaptoge S, et al. Lipo-protein(a) concentration and the risk of coronary heart disease, stroke, and nonvascular mortality. JAMA 2009;302:412–23.
- Madsen CM, Kamstrup PR, Langsted A, Varbo A, Nordestgaard BG. Lipoprotein(a)-lowering by 50 mg/dL (105 nmol/L) may be needed to reduce cardiovascular disease 20% in secondary prevention: a population-based study. Arterioscler Thromb Vasc Biol 2020;40:255–66.
- Pare G, Caku A, McQueen M, et al. Lipoprotein(a) levels and the risk of myocardial infarction among 7 ethnic groups. Circulation 2019;139:1472–82.
- Enkhmaa B, Anuurad E, Berglund L. Lipoprotein (a): impact by ethnicity and environmental and medical conditions. J Lipid Res 2016;57:1111–25.
- O’Donoghue ML, Fazio S, Giugliano RP, et al. Lipoprotein(a), PCSK9 inhibition, and cardiovascular risk. Circulation 2019;139:1483–92.
- Bittner VA, Szarek M, Aylward PE, et al. Effect of alirocumab on lipoprotein(a) and cardiovascular risk after acute coronary syndrome. J Am Coll Cardiol 2020;75:133–44.
- Wang Z, Zhai X, Xue M, Cheng W, Hu H. Prognostic value of lipoprotein (a) level in patients with coronary artery disease: a meta-analysis. Lipids Health Dis 2019;18:150.56.
- Pan Y, Li H, Meng X, Wang Y. Causal effect of Lp(a) [lipoprotein(a)] level on ischemic stroke and Alzheimer disease: a Mendelian randomization study. Stroke 2019;50:3532–9.
- Langsted A, Nordestgaard BG, Kamstrup PR. Elevated lipoprotein(a)and risk of ischemic stroke. J Am Coll Cardiol 2019;74:54–66.
- Kotani K, Sahebkar A, Serban MC, et al. Lipoprotein(a) levels inpatients with abdominal aortic aneurysm. Angiology 2017;68:99–108.
- Mortensen MB, Afzal S, Nordestgaard BG, Falk E. Primary prevention with statins: ACC/AHA risk-based approach versus trial-based approaches to guide statin therapy. J Am Coll Cardiol 2015;66:2699–709.
- Mortensen MB, Nordestgaard BG. Statin use in primary prevention of atherosclerotic cardiovascular disease according to 5 major guidelines for sensitivity, specificity, and number needed to treat. JAMA Cardiol 2019;4:1131–8.
- Mortensen MB, Falk E, Li D, et al. Statin trials, cardiovascular events, andcoronary artery calcification: implications for a trial-based approach to statin therapy in MESA. JACC Cardiovasc Imaging 2018;11:221–30.
- Blaha MJ, Silverman MG, Budoff MJ. Is there a role for coronary artery calcium scoring for management of asymptomatic patients at risk for coronary artery disease?: Clinical risk scores are not sufficient to define primary prevention treatment strategies among asymptomatic patients. Circ Cardiovasc Imaging 2014;7:398–408. [discussion: 408].
- Silverman MG, Blaha MJ, Krumholz HM, et al. Impact of coronary artery calcium on coronary heart disease events in individuals at the extremes of traditional risk factor burden: the Multi-Ethnic Study of Atherosclerosis. Eur Heart J 2014;35:2232–41.
- Grandhi GR, Mirbolouk M, Dardari ZA, et al. Interplay of coronary artery calcium and risk factors for predicting CVD/CHD mortality: the CAC Consortium. JACC Cardiovasc Imaging 2020;13:1175–86.
- Villines TC, Hulten EA, Shaw LJ, et al. Prevalence and severity of coronary artery disease and adverse events among symptomatic patients with coronary artery calcification scores of zero undergoing coronary computed tomography angiography: results from the CONFIRM (Coronary CT Angiography Evaluation for Clinical Outcomes: An International Multicenter) registry. J Am Coll Cardiol 2011;58:2533–40.
- Orimoloye OA, Budoff MJ, Dardari ZA, et al. Race/ethnicity and the prognostic implications of coronary artery calcium for all-cause and cardiovascular disease mortality: the Coronary Artery Calcium Consortium. J Am Heart Assoc 2018;7: e010471.
- McClelland RL, Jorgensen NW, Budoff M, et al. 10-Year coronary heart disease risk prediction using coronary artery calcium and traditional risk factors: derivation in the MESA (Multi-Ethnic Study of Atherosclerosis) with validation in the HNR (Heinz Nixdorf Recall) study and the DHS (Dallas Heart Study). J Am Coll Cardiol 2015;66:1643–53.
- Peters SA, den Ruijter HM, Bots ML, Moons KG. Improvements in risk stratification for the occurrence of cardiovascular disease by imaging subclinical atherosclerosis: a systematic review. Heart 2012;98:177–84.
- Yeboah J, McClelland RL, Polonsky TS, et al. Comparison of novel risk markers for improvement in cardiovascular risk assessment in intermediate-risk individuals. JAMA 2012;308:788–95.
- Greenland P, Blaha MJ, Budoff MJ, Erbel R, Watson KE. Coronary calcium score and cardiovascular risk. J Am Coll Cardiol 2018;72:434–47.
- Mahabadi AA, Mohlenkamp S, Lehmann N, et al. CAC score improves coronary and CV risk assessment above statin indication by ESC and AHA/ACC primary prevention guidelines. JACC Cardiovasc Imaging 2017;10:143–53.
- Alashi A, Lang R, Seballos R, et al. Reclassification of coronary heart disease risk in a primary prevention setting: traditional risk factor assessment vs. coronary artery calcium scoring. Cardiovasc Diagn Ther 2019;9:214–20.
- Taylor AJ, Cerqueira M, Hodgson JM, et al. ACCF/SCCT/ACR/AHA/ASE/ASNC/NASCI/SCAI/SCMR 2010 appropriate use criteria for cardiac computed tomography. A report of the American College of Cardiology Foundation Appropriate Use Criteria Task Force, the Society of Cardiovascular Computed Tomography, the American 1146 Canadian Journal of Cardiology Volume 37 2021 College of Radiology, the American Heart Association, the American Society of Echocardiography, the American Society of Nuclear Cardiology, the North American Society for Cardiovascular Imaging, the Society for Cardiovascular Angiography and Interventions, and the Society for Cardiovascular Magnetic Resonance. J Am Coll Cardiol 2010;56:1864–94.
- Sarwar A, Shaw LJ, Shapiro MD, et al. Diagnostic and prognostic value of absence of coronary artery calcification. JACC Cardiovasc Imaging 2009;2:675–88.
- Blaha MJ, Cainzos-Achirica M, Dardari Z, et al. All-cause and cause specific mortality in individuals with zero and minimal coronary artery calcium: a long-term, competing risk analysis in the Coronary Artery Calcium Consortium. Atherosclerosis 2020;294:72–9.
- Mohlenkamp S, Lehmann N, Greenland P, et al. Coronary artery calcium score improves cardiovascular risk prediction in persons without indication for statin therapy. Atherosclerosis 2011;215:229–36.
- Budoff MJ, Young R, Burke G, et al. Ten-year association of coronary artery calcium with atherosclerotic cardiovascular disease (ASCVD) events: the multi-ethnic study of atherosclerosis (MESA). Eur Heart J 2018;39:2401–8.
- McEvoy JW, Blaha MJ, Rivera JJ, et al. Mortality rates in smokers and nonsmokers in the presence or absence of coronary artery calcification. JACC Cardiovasc Imaging 2012;5:1037–45.
- Cohen R, Budoff M, McClelland RL, et al. Significance of a positive family history for coronary heart disease in patients with a zero coronary artery calcium score (from the Multi-Ethnic Study of Atherosclerosis). Am J Cardiol 2014;114:1210–4.
- Dudum R, Dzaye O, Mirbolouk M, et al. Coronary artery calcium scoring in low risk patients with family history of coronary heart disease: validation of the SCCT guideline approach in the coronary artery calcium consortium. J Cardiovasc Comput Tomogr 2019;13:21–5.
- Min JK, Lin FY, Gidseg DS, et al. Determinants of coronary calcium conversion among patients with a normal coronary calcium scan: what is the “warranty period” for remaining normal? J Am Coll Cardiol 2010;55:1110–7.
- Rozanski A, Gransar H, Shaw LJ, et al. Impact of coronary artery calcium scanning on coronary risk factors and downstream testing the EISNER (Early Identification of Subclinical Atherosclerosis by Noninvasive Imaging Research) prospective randomized trial. J Am Coll Cardiol 2011;57:1622–32.
- Gupta A, Lau E, Varshney R, et al. The identification of calcified coronary plaque is associated with initiation and continuation of pharmaco-logical and lifestyle preventive therapies: a systematic review and meta-analysis. JACC Cardiovasc Imaging 2017;10:833–42.
- Miname MH, Bittencourt MS, Moraes SR, et al. Coronary artery calcium and cardiovascular events in patients with familial hypercholes-terolemia receiving standard lipid-lowering therapy. JACC Cardiovasc Imaging 2019;12:1797–804.
- Miname MH, Bittencourt MS, Pereira AC, et al. Vascular age derived from coronary artery calcium score on the risk stratification of individuals with heterozygous familial hypercholesterolaemia. Eur Heart J Cardiovasc Imaging 2019;21:251–7.
- Puri R, Nicholls SJ, Shao M, et al. Impact of statins on serial coronary calcification during atheroma progression and regression. J Am Coll Cardiol 2015;65:1273–82.
- Hong JC, Blankstein R, Shaw LJ, et al. Implications of coronary artery calcium testing for treatment decisions among statin candidates according to the ACC/AHA cholesterol management guidelines: a cost-effectiveness analysis. JACC Cardiovasc Imaging 2017;10:938–52.
- Ridker PM, Danielson E, Fonseca FAH, et al. Rosuvastatin to prevent vascular events in men and women with elevated C-reactive protein. NEngl J Med 2008;359:2195–207.
- Sabatine MS, Giugliano RP, Keech AC, et al. Evolocumab and clinical outcomes in patients with cardiovascular disease. N Engl J Med 2017;376:1713–22.
- Schwartz GG, Steg PG, Szarek M, et al. Alirocumab and cardiovascular outcomes after acute coronary syndrome. N Engl J Med 2018;379:2097–107.
- Raal FJ, Honarpour N, Blom DJ, et al. Inhibition of PCSK9 with evo-locumab in homozygous familial hypercholesterolaemia (TESLA Part B): a randomised, double-blind, placebo-controlled trial. Lancet 2015;385:341–50.
- Ference BA, Ginsberg HN, Graham I, et al. Low-density lipoproteins cause atherosclerotic cardiovascular disease. 1. Evidence from genetic, epidemiologic, and clinical studies. A consensus statement from the European Atherosclerosis Society Consensus Panel. Eur Heart J 2017;38:2459–72.
- Boren J, Chapman MJ, Krauss RM, et al. Low-density lipoproteins cause atherosclerotic cardiovascular disease: pathophysiological, genetic, and therapeutic insights: a consensus statement from the European Atherosclerosis Society Consensus Panel. Eur Heart J 2020;41:2313–30.
- Grundy SM, Stone NJ, Bailey AL, et al. 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA guideline on the management of blood cholesterol: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines [erratum in: 2019;139:e1182-6]. Circulation 2019;139:e1082–143.
- Cholesterol Treatment Trialists (CTT) CollaboratorsMihaylova B, Emberson J, et al. The effects of lowering LDL cholesterol with statin therapy in people at low risk of vascular disease: meta-analysis of individual data from 27 randomised trials. Lancet 2012;380:581–90.
- The HPS3/TIMI55−REVEAL Collaborative Group. Effects of anace-trapib in patients with atherosclerotic vascular disease. N Engl J Med 2017;377:1217–27.
- Scandinavian Survival Study Group. Randomised trial of cholesterol lowering in 4444 patients with coronary heart disease: the Scandinavian Simvastatin Survival Study (4S). Lancet 1994;344:1383–9.
- Cannon CP, Blazing MA, Giugliano RP, et al. Ezetimibe added to statin therapy after acute coronary syndromes. N Engl J Med 2015;372:2387–97.
- Cholesterol Treatment Trialists’ (CTT) Collaboration. Efficacy and safety of more intensive lowering of LDL cholesterol: a meta-analysis of data from 170 000 participants in 26 randomised trials. Lancet 2010;376:1670–81.
- Taguchi I, Iimuro S, Iwata H, et al. High-dose versus low-dose pitavastatin in Japanese Patients with stable coronary artery disease (REAL-CAD). A randomized superiority trial. Circulation 2018;137:1997–2009.
- Amarenco P, Kim JS, Labreuche J, et al. A comparison of two LDL cholesterol targets after ischemic stroke. N Engl J Med 2020;382:9–19.
- Kastelein JJP, Ginsberg HN, Langslet G, et al. ODYSSEY FH I and FH II: 78 week results with alirocumab treatment in 735 patients with heterozygous familial hypercholesterolaemia. Eur Heart J 2015;36:2996–3003.
- Boekholdt SM, Hovingh GK, Mora S, et al. Very low levels of athero-genic lipoproteins and the risk for cardiovascular events: a meta-analysis of statin trials. J Am Coll Cardiol 2014;64:485–94.
- Giugliano RP, Peterson TR, Park G, et al. Clinical efficacy and safety of achieving very low LDL-cholesterol concentrations with the PCSK9 inhibitor evolocumab: a prespecified secondary analysis of the Further Cardiovascular Outcomes Research with PCSK9 Inhibition in Subjects with Elevated Risk (FOURIER) trial. Lancet 2017;390:1962–71.
- Gencer B, Mach F, Murphy SA, et al. Efficacy of evolocumab on cardiovascular outcomes in patients with recent myocardial infarction: a prespecified secondary analysis from the Further Cardiovascular Outcomes Research with PCSK9 Inhibition in Subjects with Elevated Risk (FOURIER) trial. JAMA Cardiol 2020;5:1–6.
- Sabatine M, Leiter LA, Wiviott SD, et al. Cardiovascular safety and efficacy of the PCSK9 inhibitor evolocumab in patients with and without diabetes and the effect of evolocumab on glycaemia and risk of new-onset diabetes: a prespecified analysis of the Further Cardiovascular Outcomes Research with PCSK9 Inhibition in Subjects with Elevated Risk (FOURIER) randomized controlled trial. Lancet Diabetes Endocrinol 2017;5:941–50.
- Deedwania P, Murphy SA, Scheen A, et al. Efficacy and safety of PCSK9 inhibition with evolocumab in reducing cardiovascular events in patients with metabolic syndrome receiving statin therapy: secondary analysis from the Further Cardiovascular Outcomes Research with PCSK9 Inhibition in Subjects with Elevated Risk (FOURIER) randomized clinical trial. JAMA Cardiol 2021;6:139–47.
- Ray K, Colhoun HM, Szarek M, et al. Effects of alirocumab on cardiovascular and metabolic outcomes after acute coronary syndrome in patients with or without diabetes: a prespecified analysis of the ODYSSEY OUTCOMES randomised controlled trial. Lancet Diabetes Endocrinol 2019;7:618–28.
- Bonaca MP, Nault P, Giugliano RP, et al. Low-density lipoprotein cholesterol lowering with evolocumab and outcomes in patients with peripheral artery disease. Insights from the Further Cardiovascular Outcomes Research with PCSK9 Inhibition in Subjects with Elevated Risk (FOURIER). Trial. Circulation 2018;137:338–50.
- Jukkema JW, Szarek M, Zijlstra. Alirocumab in patients with polyvascular disease and recent acute coronary syndrome. ODYSSEY OUTCOMES Trial. J Am Coll Cardiol 2019;74:1167–76.
- Sabatine MS, De Ferrari GM, Giugliano RP, et al. Clinical benefit of evolocumab by severity and extent of coronary artery disease: analysis from Further Cardiovascular Outcomes Research with PCSK9 Inhibition in Subjects with Elevated Risk (FOURIER). Circulation 2018;138:756–66.
- Goodman SG, Aylward PE, Szarek M. Effects of alirocumab on cardiovascular events after coronary bypass surgery. J Am Coll Cardiol 2019;74:1177–86.
- Bittner VA, Szarek M, Aylward PE. Effect of alirocumab on lipoprotein (a) and cardiovascular risk after acute coronary syndrome. J Am Coll Cardiol 2020;75:133–44.
- Bhatt DL, Steg PG, Miller M, et al. Cardiovascular risk reduction with icosapent ethyl for hypertriglyceridemia. N Engl J Med 2019;380:11–22.
- Robinson JG, Rosenson RS, Farnier M, et al. Safety of very low low-density lipoprotein cholesterol levels with alirocumab: pooled data from randomized trials. J Am Coll Cardiol 2017;69:471–82.
- Iqbal Z, Dhage S, Mohamad JB. Efficacy and safety of PCSK9 monoclonal antibodies. Expert Opin Drug Saf 2019;18:1191–201.
- Hagiwara N, Kawada-Watanabe E, Koyanagi R, et al. Low-density lipo-protein cholesterol targeting with pitavastatin + ezetimibe for patients with acute coronary syndrome and dyslipidaemia: the HIJ-PROPER study, a prospective, open-label, randomized trial. Eur Heart J 2017;38:2264–75.
- Alkindi M, Siminovitch KA, Gupta M, Genest J. Monoclonal antibodies for the treatment of hypercholesterolemia: targeting PCSK9. Can J Cardiol 2016;32:1552–60.
- Turgeon RD, Tsuyuki RT, Gyenes GT, Pearson GJ. Cardiovascular efficacy and safety of PCSK9 inhibitors: systematic review and meta-analysis including the ODYSSEY OUTCOMES Trial. Can J Cardiol 2018;34:1600–5.
- Moriarty PM, Thompson PD, Cannon CP, et al. Efficacy and safety of alirocumab vs ezetimibe in statin-intolerant patients, with a statin rechallenge arm: The ODYSSEY ALTERNATIVE randomized trial. J Clin Lipidol 2015;9:758–69.
- Nissen SE, Stroes E, Dent-Acosta RE, et al. Efficacy and tolerability of evolocumab vs ezetimibe in patients with muscle-related statin intolerance: the GAUSS-3 randomized clinical trial. JAMA 2016;315:1580–90.
- Raal FJ, Stein EA, Dufour R, et al. PCSK9 inhibition with evolocumab (AMG 145) in heterozygous familial hypercholesterolaemia (RUTHERFORD-2): a randomised, double-blind, placebo-controlled trial. Lancet 2015;385:331–40.
- ASCEND Study Collaborative Group. Effects of n-3 fatty acid supplements in diabetes mellitus. N Engl J Med 2018;379:1540–50.
- Manson JE, Cook NR, Lee IM, et al. Marine n-3 fatty acids and prevention of cardiovascular disease and cancer. N Engl J Med 2019;380:23–32.
- Yokoyama M, Origasa H, Matsuzaki M, et al. Effects of eicosapentaenoic acid on major coronary events in hypercholesterolaemic patients (JELIS): a randomised open-label, blinded endpoint analysis. Lancet 2007;369:1090–8.
- Nicholls SJ, Lincoff AM, Garcia M, et al. Effect of high-dose omega-3 fatty acids vs corn oil on major adverse cardiovascular events in patients at high cardiovascular risk: the STRENGTH randomized clinical trial. JAMA 2020;324:2268–80.
- ClinicalTrials.gov. Effect of evolocumab in patients at high cardiovascular risk without prior myocardial infarction or stroke (VERSALIUS-CV). Available at: https://clinicaltrials.gov/ct2/show/NCT03872401. Accessed September 3, 2020.
- Raal FJ, Kallend D, Ray KK, et al. Inclisiran for the treatment of heterozygous familial hypercholesterolemia. N Engl J Med 2020;382:1520–30.
- Ray KK, Wright RS, Kallend D, et al. Two phase 3 trials of inclisiran in patients with elevated LDL cholesterol. N Engl J Med 2020;382:1507–19.
- ClinicalTrials.gov. A randomized trial assessing the effects of inclisiran on clinical outcomes among people with cardiovascular disease (ORION-4). Available at: https://clinicaltrials.gov/ct2/show/ NCT03705234. Accessed September 3, 2020.
- ClincalTrials.gov. Effect of dalcetrapib vs placebo on CV risk on a genetically defined population with a recent ACS (dal-GenE). Available at: https://clinicaltrials.gov/ct2/show/NCT02525939. Accessed September 3, 2020.
- ClinicalTrials.gov. Evaluation of major cardiovascular events in patients with, or at high risk for, cardiovascular disease who are statin intolerant treated with bempedoic acid (ETC-1002) or placebo (CLEAR Outcomes). Available at: https://clinicaltrials.gov/ct2/show/ NCT02993406. Accessed September 3, 2020.
- ClinicalTrials.gov. Pemafibrate to reduce cardiovascular outcomes by reducing triglycerides in patients with diabetes (PROMINENT). Available at: https://clinicaltrials.gov/ct2/show/NCT03071692. Accessed September 3, 2020.
- Brunham LR, Ruel I, Aljenedil S, et al. Canadian Cardiovascular Society position statement on familial hypercholesterolemia: update 2018. Can J Cardiol 2018;34:1553–63.
- ASCEND Study Collaborative GroupBowman L, Mafham M, et al. Effects of n-3 fatty acid supplements in diabetes mellitus. N Engl J Med 2018;9:1540–50.
- Manson JE, Cook NR, Lee IM, et al. Marine n-3 fatty acids and prevention of cardiovascular disease and cancer. N Engl J Med 2019;380:23–32.
- Abdelhamid AS, Brown TJ, Brainard JS, et al. Omega-3 fatty acids for the primary and secondary prevention of cardiovascular disease. Cochrane Database Syst Rev 2018;11: CD003177.
- Bernstein AM, Ding EL, Willett WC, Rimm EB. A meta-analysis shows that docosahexaenoic acid from algal oil reduces serum triglycerides and increases HDL-cholesterol and LDL-cholesterol in persons without coronary heart disease. J Nutr 2012;142:99–104.
- Eslick GD, Howe PR, Smith C, Priest R, Bensoussan A. Benefits of fish oil supplementation in hyperlipidemia: a systematic review and meta-analysis. Int J Cardiol 2009;136:4–16.
- Balk EM, Lichtenstein AH, Chung M, et al. Effects of omega-3 fatty acids on serum markers of cardiovascular disease risk: a systematic review. Atherosclerosis 2006;189:19–30.
- ORIGIN Trial InvestigatorsBosch J, Gerstein HC, et al. N-3 fatty acids and cardiovascular outcomes in patients with dysglycemia. N Engl J Med 2012;367:309–18.
